Scientists just found a hidden reason inner ear cells die. A common antibiotic class is directly implicated. And the mechanism has been hiding in plain sight for years.
Why it matters
Aminoglycoside antibiotics — prescribed for drug-resistant TB to hospital-acquired infections — are activating a newly discovered cell-killing mechanism in your inner ear, causing permanent hearing loss. The tiny sensory cells, called hair cells, do not regenerate. Once they're gone, they're gone.
What we thought we knew
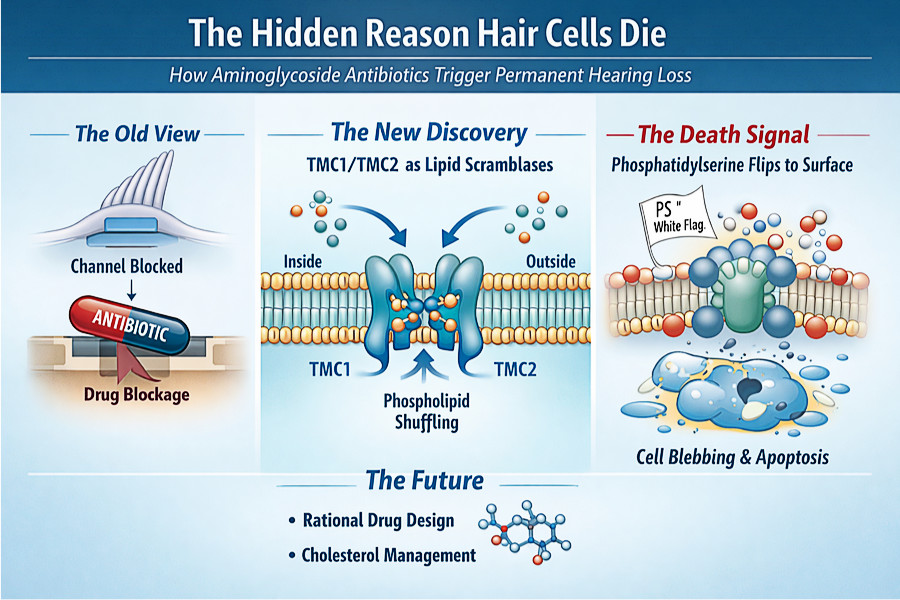
For years, scientists knew two proteins — TMC1 and TMC2 — as the machinery behind one of the body's most elegant tricks. Tiny hair-like structures, bundled like a mohawk, bend in response to sound and trigger the electrical signal your brain reads as noise. Mutations in TMC1 are already a leading cause of genetic deafness. Researchers thought they understood these proteins.
They didn't.
Assumptions, assumptions
Scientists previously believed aminoglycoside antibiotics damaged hearing by blocking TMC channel function. It was straightforward and mechanical.
Wrong.
In living hair cells, these drugs don't just block a channel. They trigger a full collapse of membrane asymmetry, activating a separate, previously unknown protein function, destroying structural order, and killing the cell. The researchers only caught this because the effect disappears entirely when you test the protein in isolation. Something in the living cellular environment — missing protein partners, specific lipids, cholesterol levels — is the actual driver.
That distinction is everything for drug designers.
A closer look
The NIDCD team at NIH discovered that TMC1 and TMC2 have a second, entirely separate job. They act as lipid scramblases, molecular machines that shuffle fatty molecules called phospholipids across cell membranes.
Here's the clinical consequence:
-
Cell membranes keep specific phospholipids on specific sides. Order is everything.
-
When that order breaks down, a phospholipid called phosphatidylserine flips to the outer membrane surface. Think of it as the cell raising a white flag, visible only when something has gone badly wrong.
-
That flip is a death signal. The cell starts blebbing. It falls apart.
"This is an apoptotic hallmark. It's what's killing the hair cells." —Angela Ballesteros, the lab's principal investigator
The intrigue
Membrane cholesterol directly modulates scramblase activity. That finding means the mechanism isn't locked — it responds to its environment. Researchers suggest diet or cholesterol management could someday act as a protective buffer against both ototoxic medications and genetic hearing loss. A systemic intervention for what has always seemed like a purely structural problem.
The takeaway
-
Drug designers now have a rational target: aminoglycoside analogs that don't activate the scramblase.
-
Genetic deafness from TMC1 mutations and drug-induced hearing loss share the same apoptotic mechanism. One discovery, two problems addressed.
-
All findings come from mouse models. No human protocols exist yet.
The drug that saves you in the ICU shouldn't cost you your hearing for life. Now researchers have a roadmap to make sure it doesn't.


